Your Cart is Empty
Watch this video and learn how to use the Good Morning Snore Solution® tongue stabilizing (retaining) device.
Finding the right solution for your personal health and wellbeing is always our number one priority. When you compare Good Morning Snore Solution to other anti-snoring devices on the market, making the right decision is easy. Get the best tongue stabilizing (retaining) device on the market today.

The simple, effective solution to breathe easy, rest easy and sleep sound. We guarantee it.
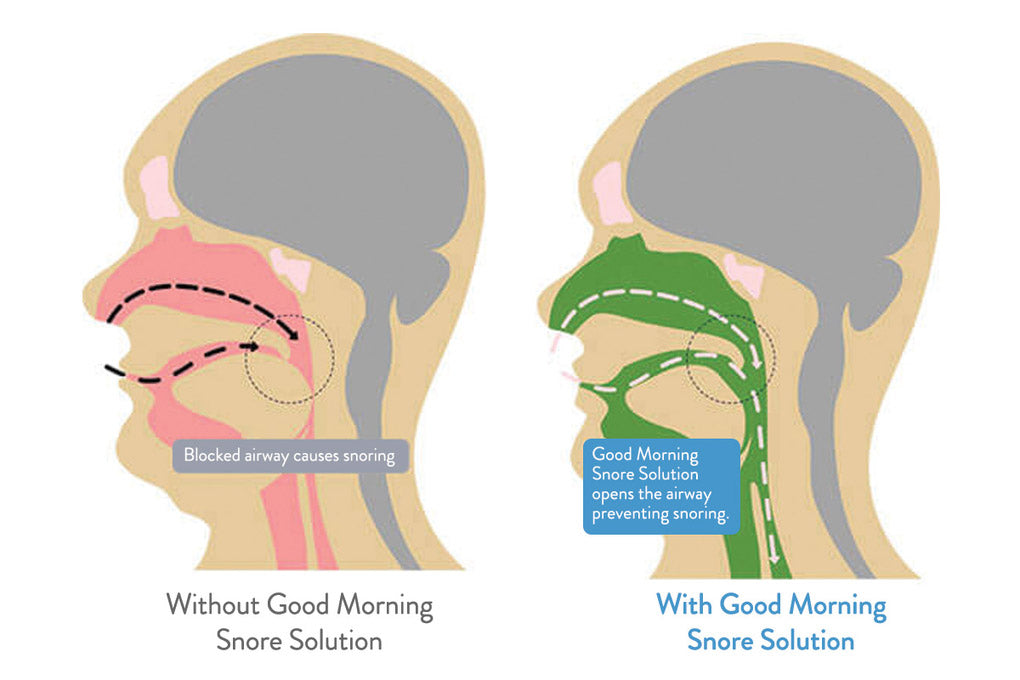
The Good Morning Snore Solution® mouthpiece uses an ingenious new method called Tongue Displacement to gently open the entire airway with few (if any) side effects.
Unlike other anti-snoring methods, Tongue Stabilization (Retaining) does not typically contribute to jaw pain, user discomfort or long-term bite misalignment.
Soft, flexible and easy to manage, the Good Morning Snore Solution® mouthpiece is simply all you need for a quiet, snore-free sleeping life.
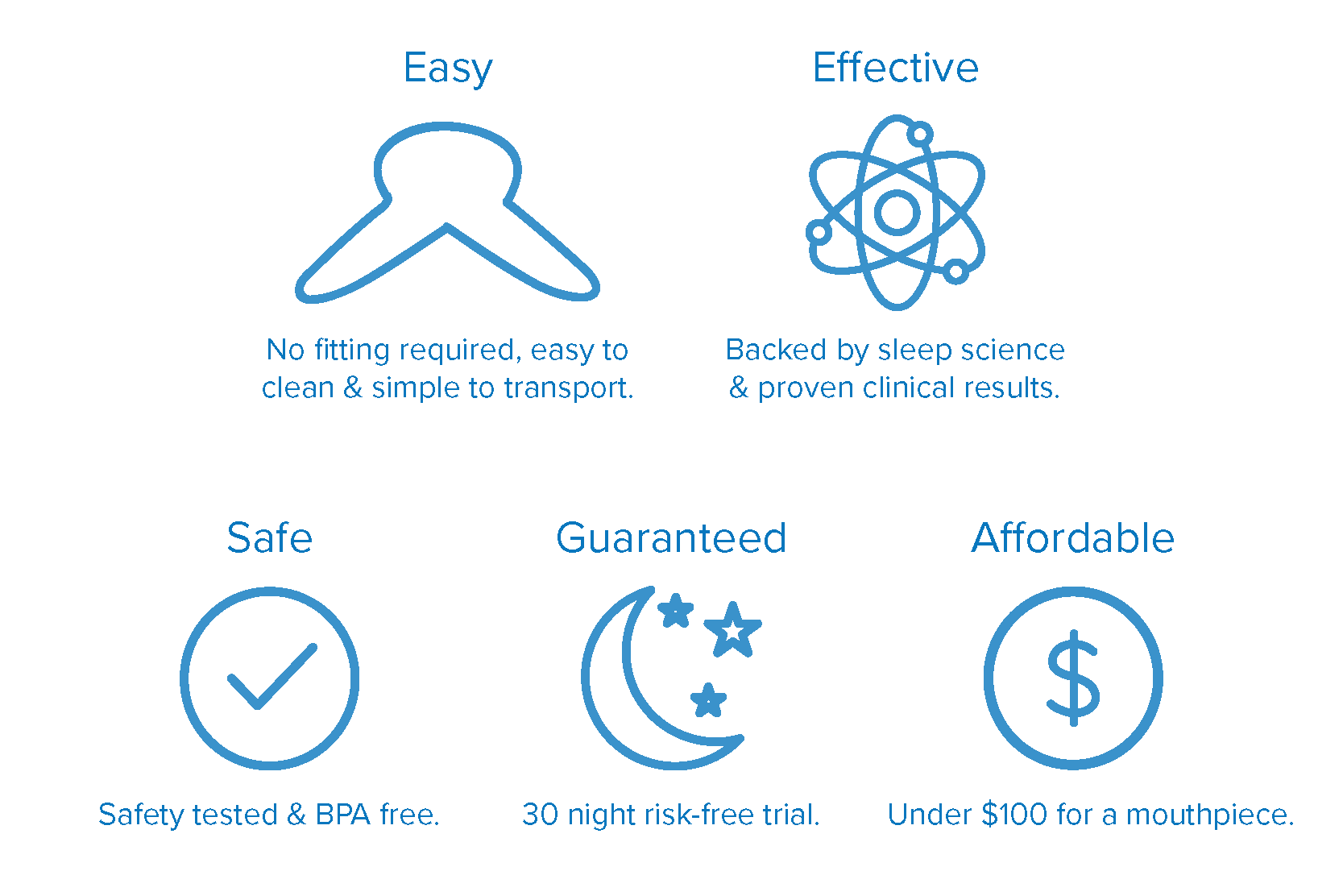
The Good Morning Snore Solution® mouthpiece is reassuringly soft, easy to use and designed to fit almost everyone.
Scientifically developed by doctors to be the most effective and least invasive solution.
Every Good Morning Snore Solution® mouthpiece comes with a complete 30-Day Satisfaction Guarantee.
